Mechanism and Experimental Study on Micro-arc Plasma Discharge Polishing of 304 Stainless Steel
-
摘要: 针对目前用于抛光不锈钢的方法普遍存在抛光质量差、抛光效率低以及环境污染严重的问题,本文以304不锈钢为加工对象,将电化学加工与放电加工相结合,提出了微弧等离子体放电抛光方法。基于电化学理论和流注理论对微弧等离子体放电抛光机理进行了研究。通过设计正交试验并采用田口分析法和方差分析法对试验结果进行了分析,找到了关于材料去除率和表面粗糙度的优化参数组合并进行了试验验证。结果表明,电源电压、抛光时间和电解液温度对材料去除率具有显著影响,抛光时间和电源电压对表面粗糙度具有显著影响。微弧等离子体放电抛光304不锈钢的材料去除率最大可以达到11.73 μm/min,表面粗糙度最小可以达到29.1 nm。Abstract: For the poor polishing quality, low polishing efficiency and serious environmental pollution in polishing of stainless steel via current methods, the micro-arc plasma discharge polishing method in the polishing of 304 stainless steel was proposed by combining the electrochemical processing with electrical discharge machining. The mechanism in the micro-arc plasma discharge polishing was studied based on the electrochemical and streamer theories. By planning the orthogonal tests and analyzing the testing results with Taguchi analysis and ANOVA, the optimal parameters by considering the material removal rate and surface roughness was obtained and verified through the experimental results. The results show that the voltage, polishing time and electrolyte temperature have the significant effects on the material removal rate, and the polishing time and voltage have the significant effects on the surface roughness. The maximum material removal rate in the micro-arc plasma discharge polishing of 304 stainless steel can reach 11.73 μm/min, and the minimum surface roughness can reach 29.1 nm.
-
Key words:
- 304 stainless steel /
- plasma /
- polishing mechanism /
- surface roughness
-
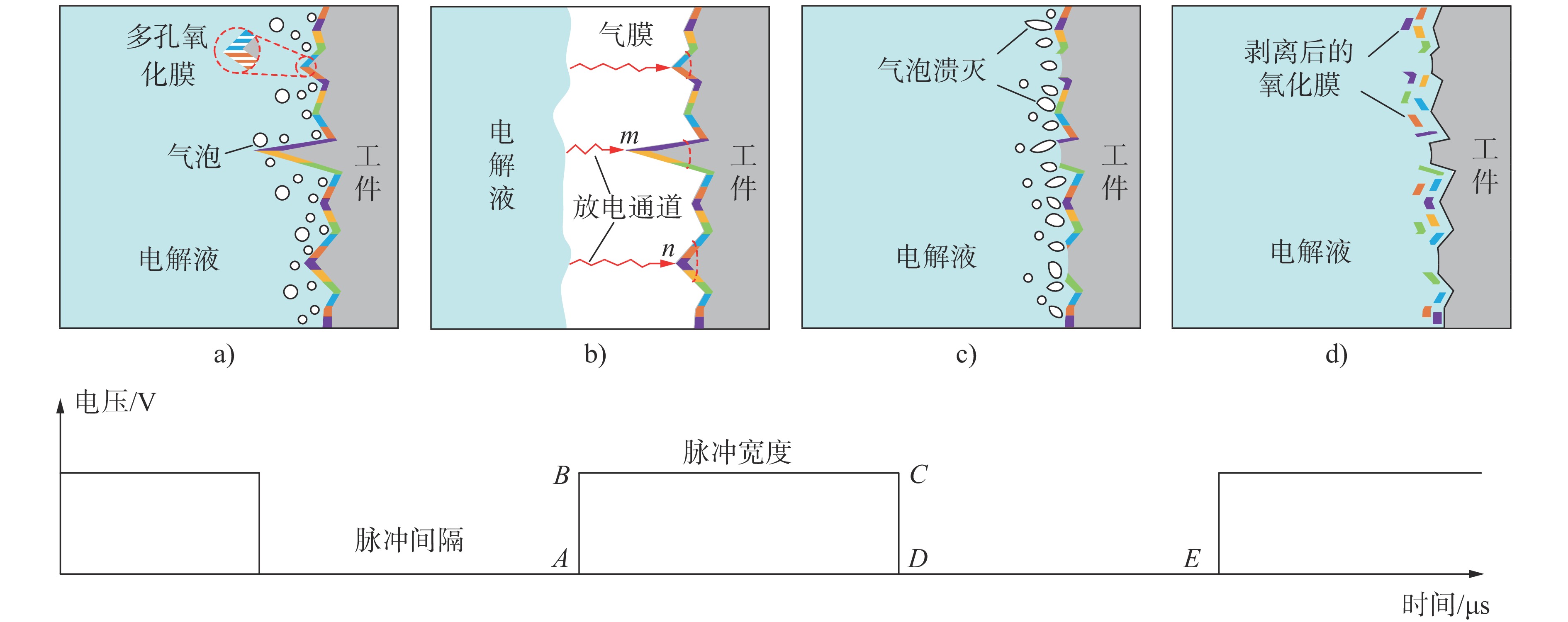
图 3 等离子体放电通道的产生过程[12]
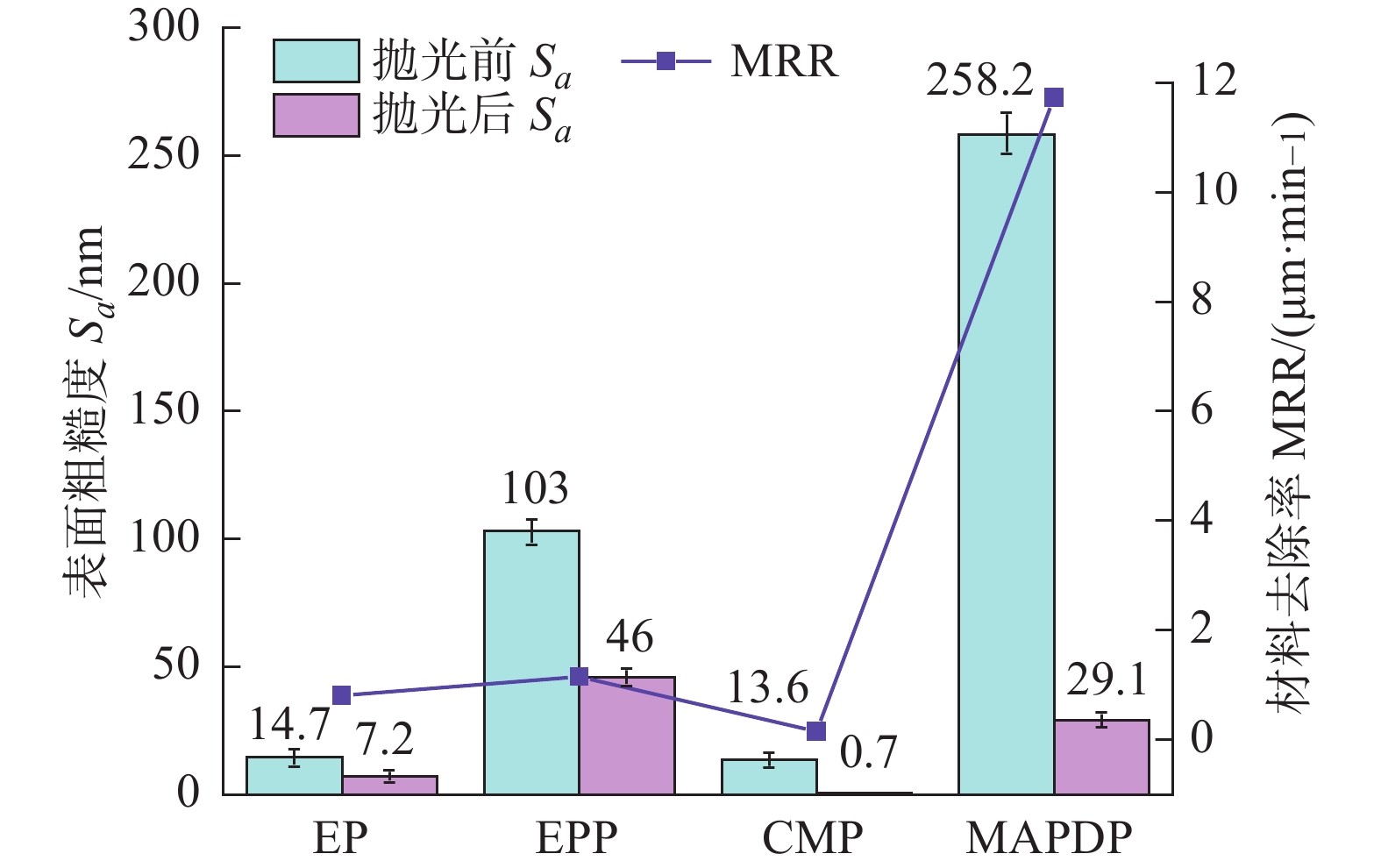
图 8 不同抛光方法的对比图[15]
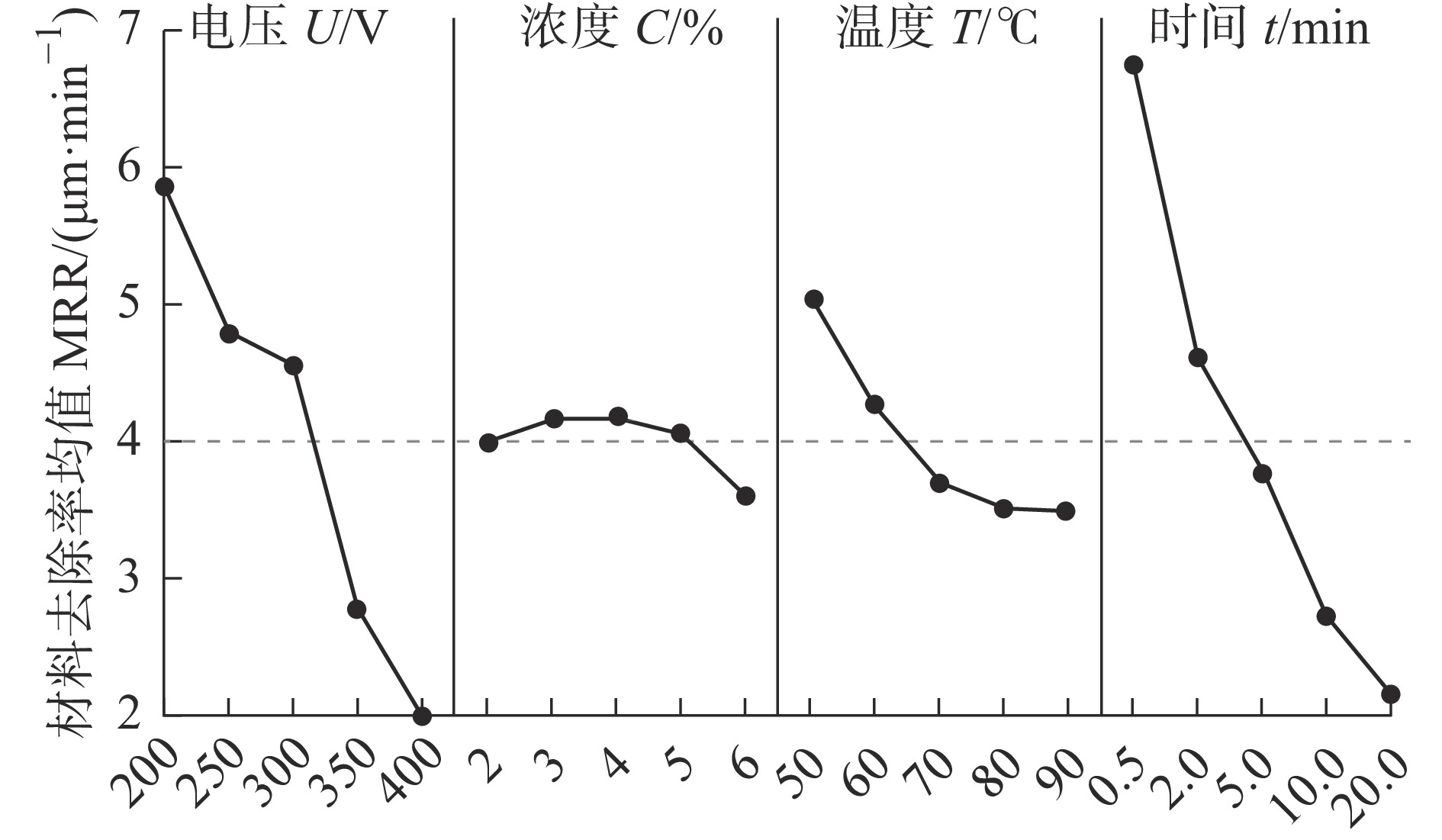
表 1 试验因素及水平
因素 水平 1 2 3 4 5 电源电压U/V 200 250 300 350 400 电解液浓度C/% 2 3 4 5 6 电解液温度T/℃ 50 60 70 80 90 抛光时间t/min 0.5 2 5 10 20 注:电解液浓度即为电解液质量分数,下同。 表 2 试验数据及结果
序号 U/V C/% T/℃ t/min Sa/μm MRR/(μm·min−1) 1 200 2 50 0.5 0.143 10.07 2 200 3 60 2 0.072 7.57 3 200 4 70 5 0.060 4.88 4 200 5 80 10 0.056 3.78 5 200 6 90 20 0.059 2.90 6 250 2 60 5 0.043 4.37 7 250 3 70 10 0.045 3.78 8 250 4 80 20 0.057 3.03 9 250 5 90 0.5 0.149 6.72 10 250 6 50 2 0.080 5.89 11 300 2 70 20 0.039 2.44 12 300 3 80 0.5 0.129 6.73 13 300 4 90 2 0.076 5.04 14 300 5 50 5 0.043 5.88 15 300 6 60 10 0.032 2.52 16 350 2 80 2 0.075 2.31 17 350 3 90 5 0.083 2.02 18 350 4 50 10 0.035 2.69 19 350 5 60 20 0.041 1.64 20 350 6 70 0.5 0.137 5.04 21 400 2 90 10 0.085 0.56 22 400 3 50 20 0.042 0.51 23 400 4 60 0.5 0.140 5.04 24 400 5 70 2 0.114 2.10 25 400 6 80 5 0.125 1.51 表 3 MRR的均值响应表
水平 电压U/V 浓度C/% 温度T/℃ 时间t/min 1 5.840 3.950 5.008 6.720 2 4.758 4.122 4.228 4.582 3 4.522 4.136 3.648 3.732 4 2.740 4.024 3.472 2.666 5 1.944 3.572 3.448 2.104 Δ 3.896 0.564 1.560 4.616 排秩 2 4 3 1 表 4 MRR的方差分析结果
因素 自由度 平方和 均方和 F值 P值 贡献率/% 电压 4 50.199 12.5496 22.57 0.000 38.49 浓度 4 1.060 0.2649 0.48 0.753 0.81 温度 4 8.839 2.2097 3.97 0.046 6.78 时间 4 65.878 16.4695 29.62 0.000 50.51 误差 8 4.448 0.5560 3.41 合计 24 100 表 5 Sa的均值响应表
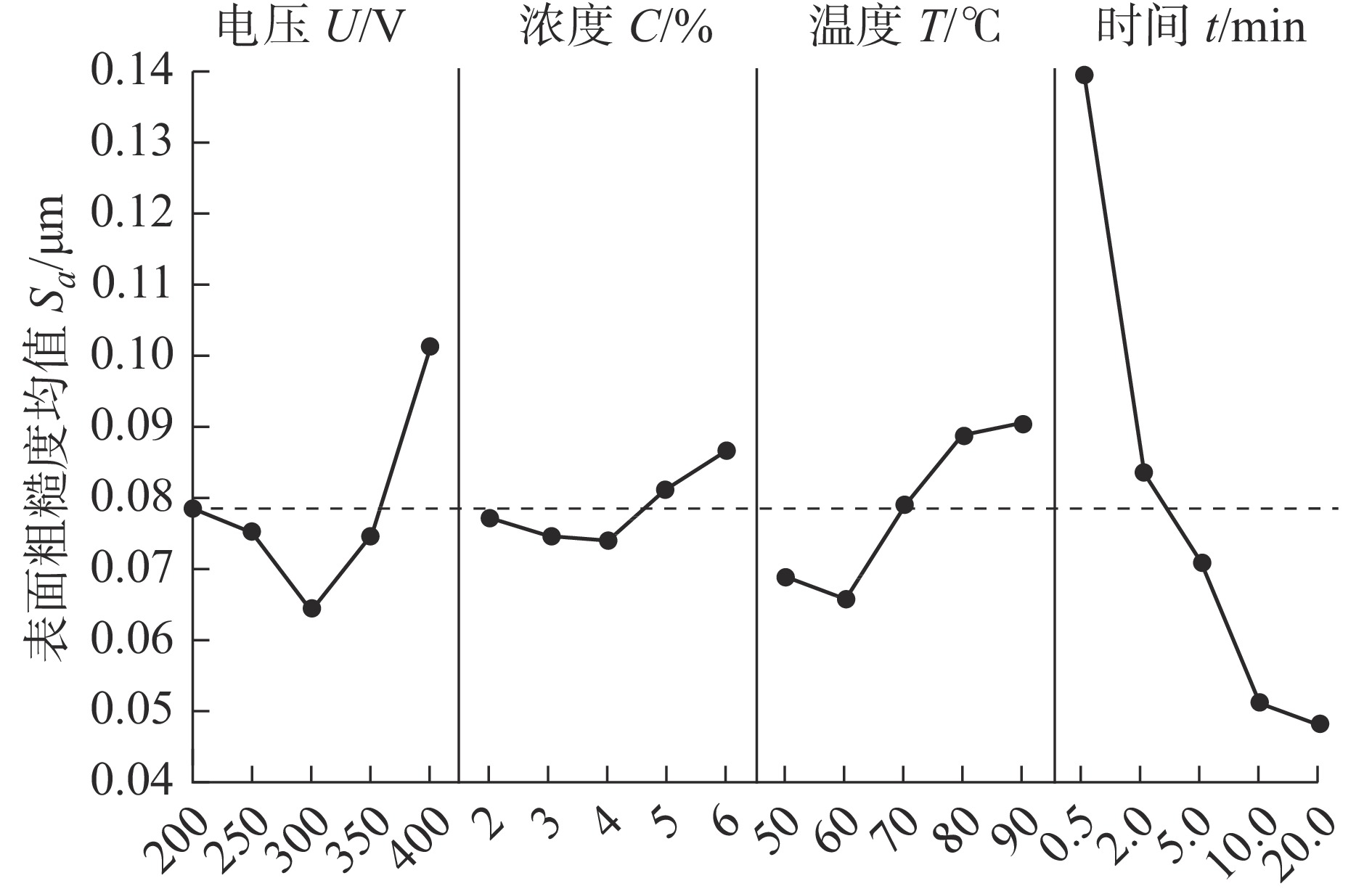
水平 电压U/V 浓度C/% 温度T/℃ 时间t/min 1 0.07800 0.07700 0.06860 0.13960 2 0.07480 0.07420 0.06560 0.08340 3 0.06380 0.07360 0.07900 0.07080 4 0.07420 0.08060 0.08840 0.05060 5 0.10120 0.08660 0.09040 0.04760 Δ 0.03740 0.01300 0.02480 0.09200 排秩 2 4 3 1 表 6 Sa的方差分析结果
因素 自由度 平方和 均方和 F值 P值 贡献率/% 电压 4 0.003819 0.000955 5.24 0.023 10.57 浓度 4 0.000574 0.000143 0.79 0.565 1.59 温度 4 0.002521 0.000630 3.46 0.064 6.98 时间 4 0.027748 0.006937 38.06 0.000 76.82 误差 8 0.001458 0.000182 4.04 合计 24 100 表 7 试验验证条件及结果
U/V C/% T/℃ t/min MRR/(μm·min−1) Sa/nm 200 4 50 0.5 11.73 300 4 60 20 29.1 -
[1] ZHANG D H, MENG X C, ZUO G Z, et al. Study of the corrosion characteristics of 304 and 316L stainless steel in the static liquid lithium[J]. Journal of Nuclear Materials, 2021, 553: 153032. doi: 10.1016/j.jnucmat.2021.153032 [2] 马幸申, 陈建钧. 奥氏体不锈钢氢损伤表面波检测的有限元模拟[J]. 机械科学与技术, 2018, 37(3): 461-465. doi: 10.13433/j.cnki.1003-8728.2018.0321MA X S, CHEN J J. Finite element simulation of surface wave test for hydrogen damage of austenite stainless steel[J]. Mechanical Science and Technology for Aerospace Engineering, 2018, 37(3): 461-465. (in Chinese) doi: 10.13433/j.cnki.1003-8728.2018.0321 [3] KAO P S, HOCHENG H. Optimization of electrochemical polishing of stainless steel by grey relational analysis[J]. Journal of Materials Processing Technology, 2003, 140(1-3): 255-259. doi: 10.1016/S0924-0136(03)00747-7 [4] ANDRADE L S, XAVIER S C, ROCHA-FILHO R C, et al. Electropolishing of AISI-304 stainless steel using an oxidizing solution originally used for electrochemical coloration[J]. Electrochimica Acta, 2005, 50(13): 2623-2627. doi: 10.1016/j.electacta.2004.11.007 [5] LIN C C, HU C C. Electropolishing of 304 stainless steel: Surface roughness control using experimental design strategies and a summarized electropolishing model[J]. Electrochimica Acta, 2008, 53(8): 3356-3363. doi: 10.1016/j.electacta.2007.11.075 [6] MAI T A, LIM G C. Micromelting and its effects on surface topography and properties in laser polishing of stainless steel[J]. Journal of Laser Applications, 2004, 16(4): 221-228. doi: 10.2351/1.1809637 [7] KANG D, ZOU P, WU H, et al. Research on ultrasonic vibration-assisted laser polishing of the 304 stainless steel[J]. Journal of Manufacturing Processes, 2021, 62: 403-417. doi: 10.1016/j.jmapro.2020.12.009 [8] LUO J F, DORNFELD D A. Material removal mechanism in chemical mechanical polishing: theory and modeling[J]. IEEE Transactions on Semiconductor Manufacturing, 2001, 14(2): 112-133. doi: 10.1109/66.920723 [9] ZHANG Z Y, LIAO L X, WANG X Z, et al. Development of a novel chemical mechanical polishing slurry and its polishing mechanisms on a nickel alloy[J]. Applied Surface Science, 2020, 506: 144670. doi: 10.1016/j.apsusc.2019.144670 [10] HU X K, SONG Z T, LIU W L, et al. Chemical mechanical polishing of stainless steel foil as flexible substrate[J]. Applied Surface Science, 2012, 258(15): 5798-5802. doi: 10.1016/j.apsusc.2012.02.100 [11] DENG T T, ZHENG Z Z, LI J J, et al. Surface polishing of AISI 304 stainless steel with micro plasma beam irradiation[J]. Applied Surface Science, 2019, 476: 796-805. doi: 10.1016/j.apsusc.2019.01.173 [12] 邵涛, 严萍. 大气压气体放电及其等离子体应用[M]. 北京: 科学出版社, 2015SHAO T, YAN P. Atmospheric pressure gas discharge and its plasma application[M]. Beijing: Science Press, 2015. (in Chinese) [13] WANG J, SUO L C, GUAN L L, et al. Analytical study on mechanism of electrolysis and plasma polishing[J]. Advanced Materials Research, 2012, 472-475: 350-353. doi: 10.4028/www.scientific.net/AMR.472-475.350 [14] SHIMA A, TAKAYAMA K, TOMITA Y, et al. Mechanism of impact pressure generation from spark-generated bubble collapse near a wall[J]. AIAA Journal, 1983, 21(1): 55-59. doi: 10.2514/3.8027 [15] JI G Q, SUN H W, DUAN H D, et al. Effect of electrolytic plasma polishing on microstructural evolution and tensile properties of 316L stainless steel[J]. Surface and Coatings Technology, 2021, 420: 127330. doi: 10.1016/j.surfcoat.2021.127330 -







 下载:
下载: